Catalyst design
Catalysis plays a key role in the conversion of fossil and renewable feedstock to high value products. The production of clean transportation fuels, of bulk and fine chemicals, and the prevention and abatement of pollution would be impossible without catalytic processes. Catalysts facilitate the formation of the desired products by selectively opening up new, energetically favourable, reaction routes. The ultimate goal for catalysis research is to design active, stable and selective catalysts.
In this research theme, we combine molecular modeling, advanced catalyst characterization, novel synthesis methods, state-of-the-art kinetic testing and microkinetic modeling to develop innovative catalytic processes up to industrial scale. To achieve breakthroughs in catalyst design, catalytic reaction mechanisms are understood and controlled at the scale of the active sites, the molecular scale. To be also industrially relevant, the mechanistic insights and design strategies are translated to and validated at the reactor scale. The close coupling between catalyst design and reactor development additionally stimulates research towards breakthrough synergies.
Our key research questions:
- What is the structure/nature of the active catalyst under reaction conditions?
- How do promotors enhance catalyst activity and selectivity?
- Can we develop design rules to control catalyst performance?
- What is the effect of a realistic feedstock on catalyst performance?
- How does the reactor choice influence the selection of optimal catalysts?
The following topics are part of the Catalyst design research theme:
Designing effective solutions to control the selectivity of catalysts
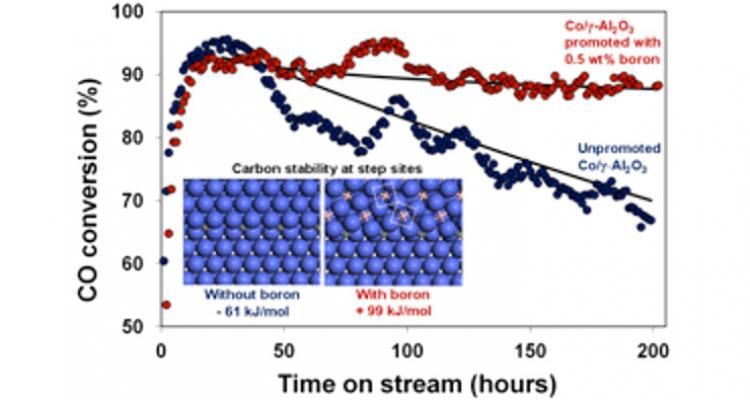
The ultimate goal for catalysis is to design catalysts with high stability, activity and selectivity. Being rather well-established for catalyst stability and activity, design principles for selectivity remain to be developed. Using first principles modeling to elucidate the catalyst structure and the nature of the active sites under reaction conditions, innovative solutions to control selectivity are sought and validated experimentally.
Analyzing complex reaction networks through transient techniques and steady-state kinetic studies
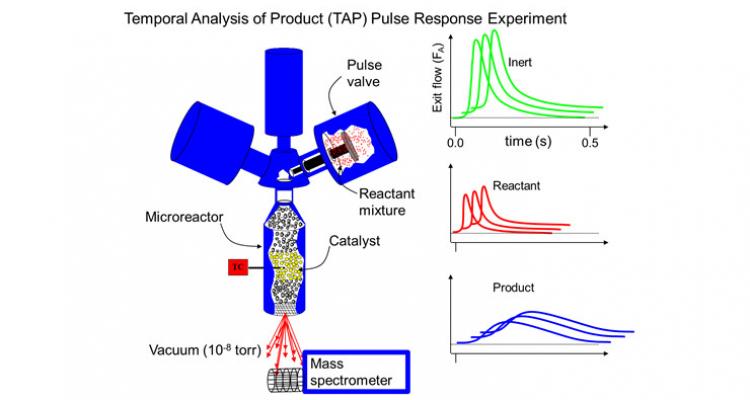
Transient techniques are particularly effective for analyzing complex reaction networks, because the rates of the various elementary steps in the catalytic cycle are not equal during transient experiments. Thus, elementary steps can be distinguished more easily, leading to a more detailed insight into the reaction kinetics. A TAP (Temporal Analysis of Products) reactor setup allows conducting a variety of transient and steady-state kinetic measurements.
Operando and in-situ material characterization
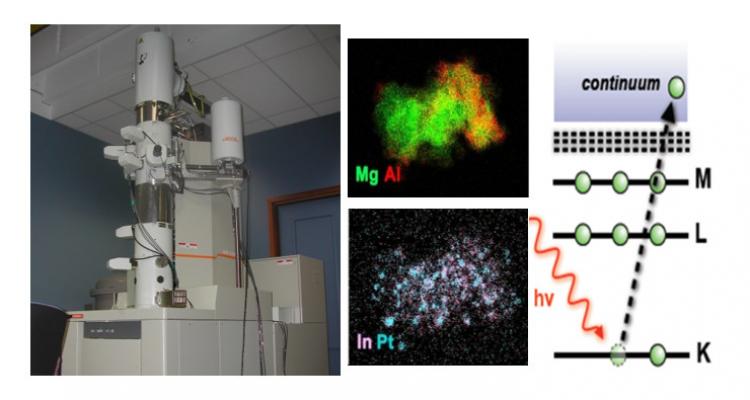
Characterization is an indispensable part of catalyst and material research. Phase, composition, elemental oxidation state, particle size and shape all join in determining the activity and stability of materials. In addition to information regarding the as-prepared and post-mortem state, an insider’s view into the material during operation is of great value. Hence the importance of operando characterization, allowing a direct connection between physico-chemical properties and performance.
Alcohol as a bio-based feedstock through optimization of the preprocessing phase

The use of alcohol, one of the prime bio-based feedstocks, requires preprocessing before it can be used as a building block in the chemical industry. The first step for the majority of processes and applications is the removal of water in catalytic dehydration. A combination of an experimental and computational approach allows to understand and optimize these catalysts.
Extending the scope and valorization of CH4
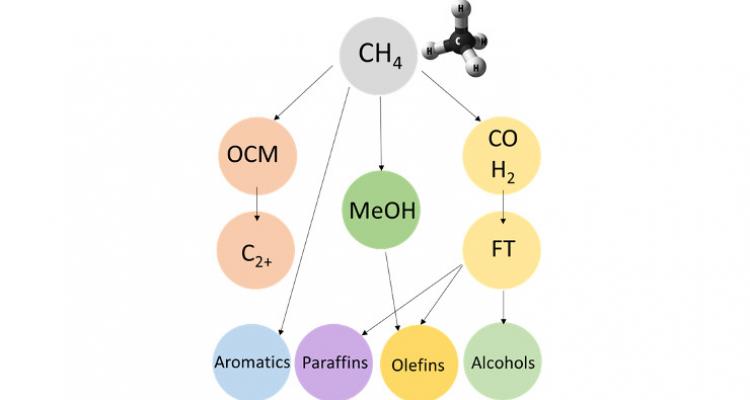
Methane activation is one of the most challenging reactions for the catalysis community. For years, most petro-chemical catalysis focused on making small building blocks out of long oil molecules. Тhis paradigm is shifting to the opposite, placing C1 catalysis at the forefront of research. It is therefore of crucial importance to further validate and extend the scope of the current C−H bond activation theories. From a materials point of view, precise control should be achieved in catalyst design and synthesis. And from a mechanistic point of view, it is imperative to develop consistent models for the understanding of catalytic methane functionalization.
Multi-scale design of reactors for catalyzed processes
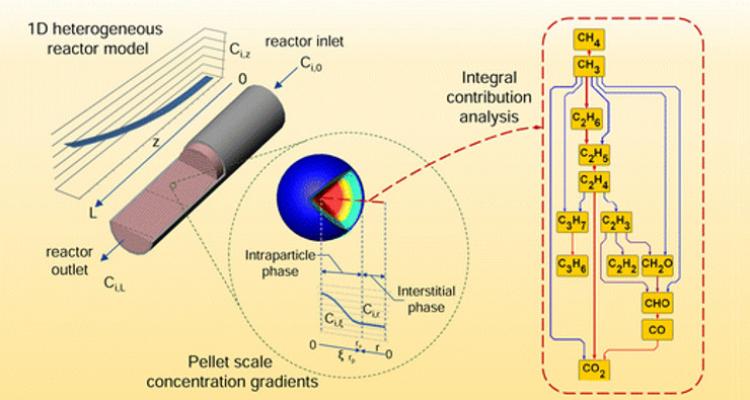
A chemical reactor is a multi-scale apparatus with occurring phenomena, ranging from the atomic scale of bond breaking and formation, over mass and heat transport at the micro/meso scale, up to the hydrodynamics at the largest scale. The ultimate challenge is to account for the elementary phenomena at these different scales to identify the dominant ones, depending on the condition used. Pushing the boundaries towards the use of microkinetic models will allow to tune the stability, activity and selectivity of the catalytic performance in these reactors.
Using hydrotreatment to upgrade conventional and renewable oil fractions and achieve cleaner emissions
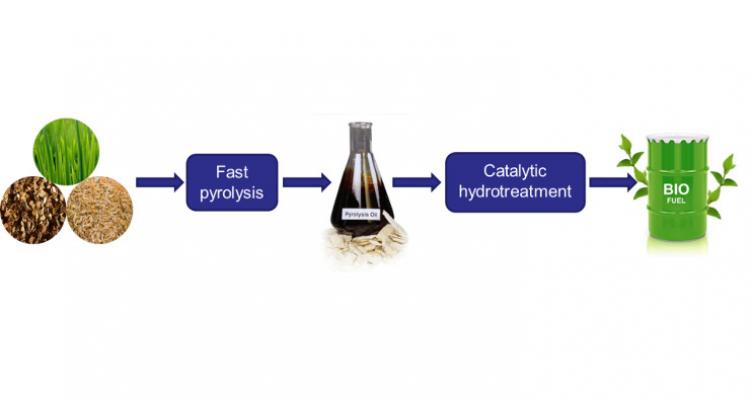
Upgrading the quality of both conventional and renewable oil fractions is frequently achieved by means of a treatment under hydrogen at elevated pressure. This reduces the unsaturation level of the feed and also leads to hetero-atom removal. Another important advantage is that cleaner emissions are achieved when using the oil products as fuel.
Metal-based catalysis for small molecule activation and selective conversion
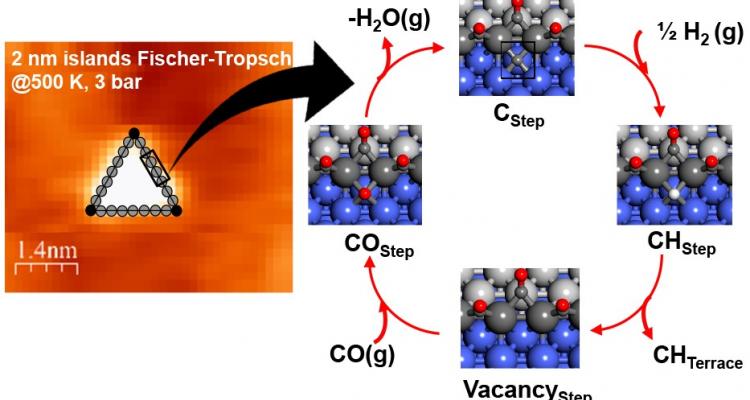
The activation and selective conversion of small molecules (e.g., CH4, CO, CO2, N2) over metal-based catalysts remains an important challenge for catalyst design and optimization. At the LCT, we combine in situ characterization, modelling-guided design and intrinsic kinetic measurements to develop novel catalysts and processes for these important transformations.
Metal catalysis to tailor the conversion of renewables into added-value products
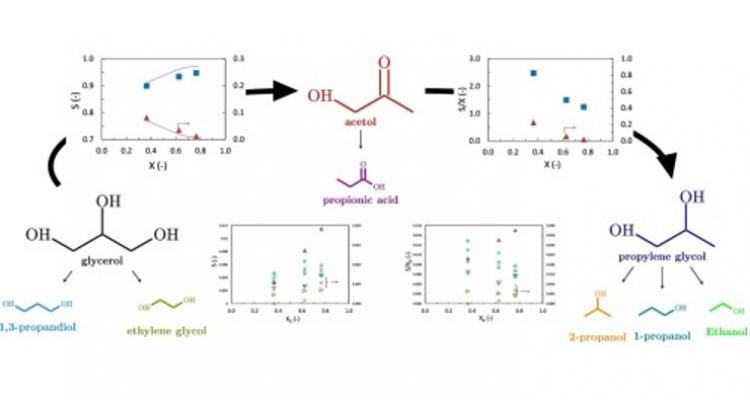
A wide range of metals can be used to efficiently catalyze the transformation of complex molecules. Activity, selectivity and stability of these catalysts are key issues to be addressed in the pursuit of optimally performing materials. Nanoparticles, alloys and specific promoting elements all open up promising opportunities to further tailor the conversion of renewables into added-value products.
Applying acid-based catalysis on renewables
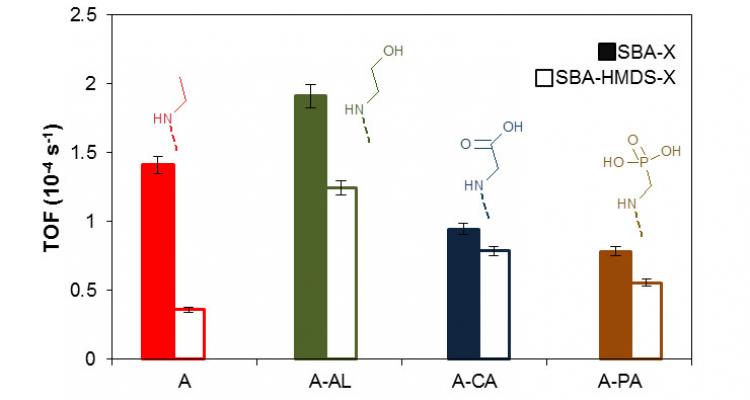
Acid catalyzed hydrocarbon conversion has a long history in refining operations and rapidly extends into the area of renewables. Complex mixtures are efficiently dealt with and - thanks to the limited number of types of elementary steps -, acid catalyzed reactions are ideally suited for fundamental kinetic modeling, such as the Single-Event MicroKinetic methodology.
Customizing the design of complex macromolecular architectures
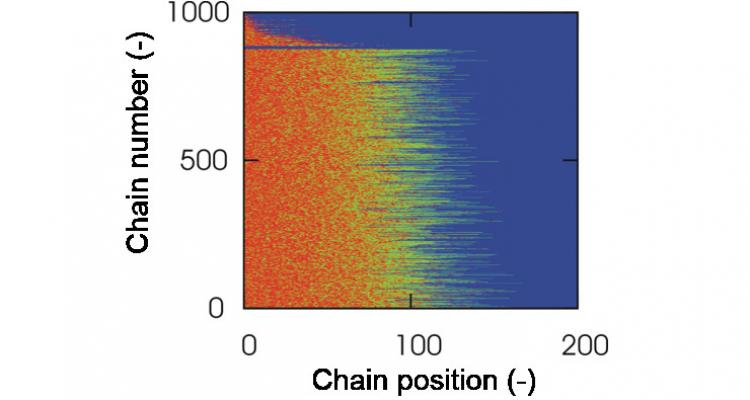
For advanced polymeric applications, such as drug delivery systems and coatings, each individual macromolecule needs to be customized with respect to its monomer sequences and location of branches. Using advanced computer algorithms, a full characterization - and thus design - of these individual macromolecules is possible.
Developing environmentally benign processes for kinetics in liquid phase
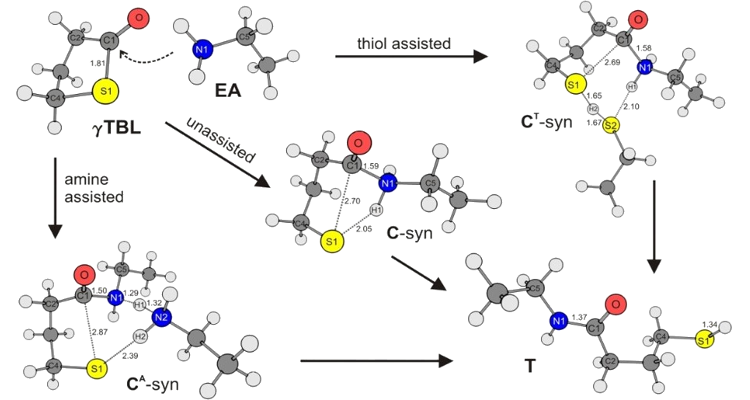
Fundamental understanding of reaction mechanisms in liquid phase - such as environmentally benign processes in the aqueous phase - requires accurate intrinsic rate coefficients. To this end, both dedicated experiments and liquid-phase computational chemistry can be applied.
Take CO2 from the atmosphere and use it as a starting material for chemical synthesis
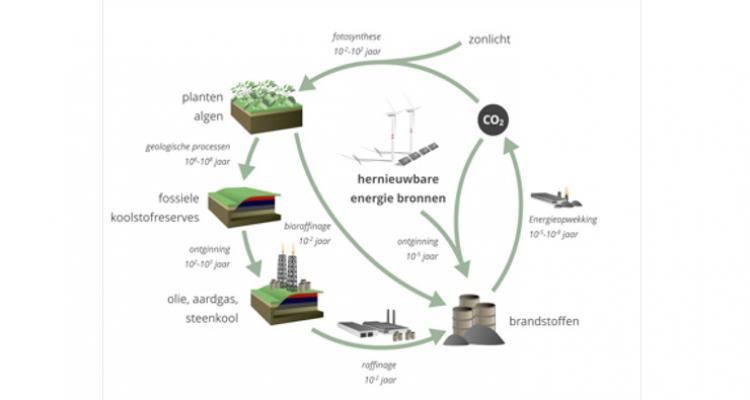
Human activities have resulted in increased concentrations of CO2 in the atmosphere, leading to climate change and acidification of the oceans. Therefore, developing efficient technologies enabling the use of CO2 as a starting material for chemical synthesis is one of the biggest scientific challenges of our time. Carbon fixation by use of CO2 as a C1 building block to replace petrochemical feedstocks is an obvious target…
