Low carbon technology
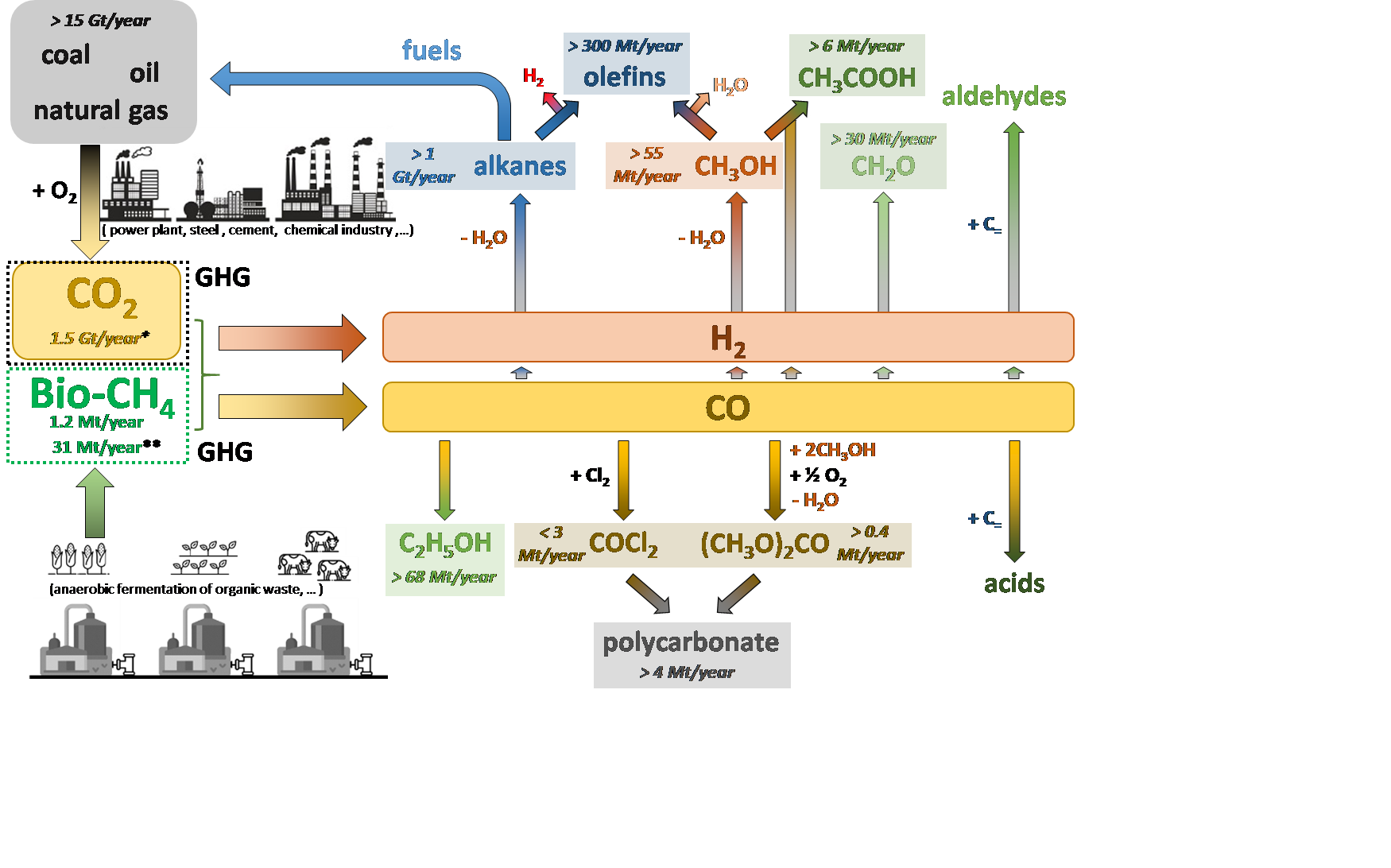
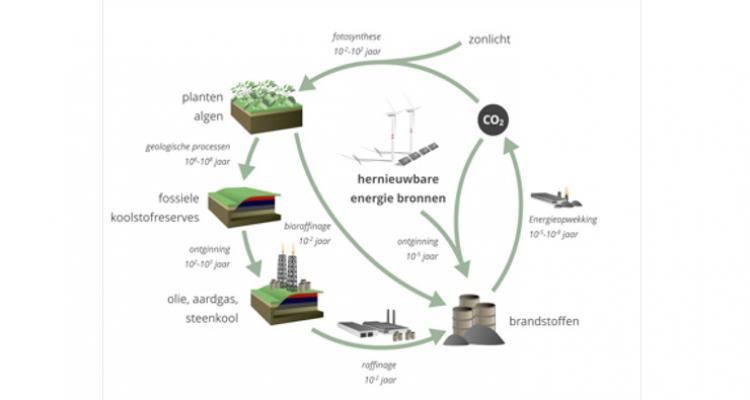
The intensive use of fossil fuels for driving industrial processes and human activities has resulted in increasingly excessive emissions of anthropogenic CO2 into the atmosphere, surpassing the 400 ppm. Being a greenhouse gas, this exceedingly high concentration of atmospheric CO2 has led to series of negative consequences for our Earth’s climate system. In efforts to mitigate the high atmospheric CO2 concentration, considerable resources have been dedicated to reduce CO2 production by replacing fossil fuels with carbon-neutral energy sources. On the other hand, CO2 serves as a C1-feedstock for the chemical industry, and thus increasing efforts are being devoted to its conversion into value-added products. The transition from a raw material base towards an almost carbon neutral economy is one of the biggest challenges. Catalysis is a key enabling factor to allow the development of new sustainable processes and technologies and thus plays a critical role to realize transition from a linear to a circular economy. Consequently, the development of catalysts and related catalytic technologies for the use of CO2 to produce chemicals and fuels, and renewable energy in chemical production is becoming a key area.
Our key research questions are:
- How can novel energy efficient processes be designed ?
- How can chemical looping provide a low carbon technology platform ?
- How can CO2 be used for the synthesis of basic chemicals ?
- To which extent can CO2, water, and renewable energy be used for synthetic fuels ?
- How can direct catalytic conversion of methane (bio-methane) into chemicals and building blocks be realized ?
- How can we contribute to the development of smartly engineered materials ?
- Can we exploit low carbon technology for energy storage ?
- Which are the tools required to provide solar-driven chemistry ?
The following topics are part of the Low carbon technology research theme:
Extending the scope and valorization of CH4
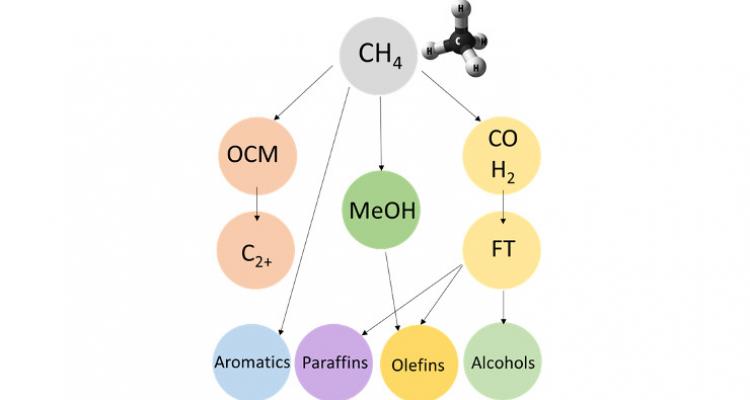
Methane activation is one of the most challenging reactions for the catalysis community. For years, most petro-chemical catalysis focused on making small building blocks out of long oil molecules. Тhis paradigm is shifting to the opposite, placing C1 catalysis at the forefront of research. It is therefore of crucial importance to further validate and extend the scope of the current C−H bond activation theories. From a materials point of view, precise control should be achieved in catalyst design and synthesis. And from a mechanistic point of view, it is imperative to develop consistent models for the understanding of catalytic methane functionalization.
Take CO2 from the atmosphere and use it as a starting material for chemical synthesis
Human activities have resulted in increased concentrations of CO2 in the atmosphere, leading to climate change and acidification of the oceans. Therefore, developing efficient technologies enabling the use of CO2 as a starting material for chemical synthesis is one of the biggest scientific challenges of our time. Carbon fixation by use of CO2 as a C1 building block to replace petrochemical feedstocks is an obvious target…
Biogas as a source for platform chemicals
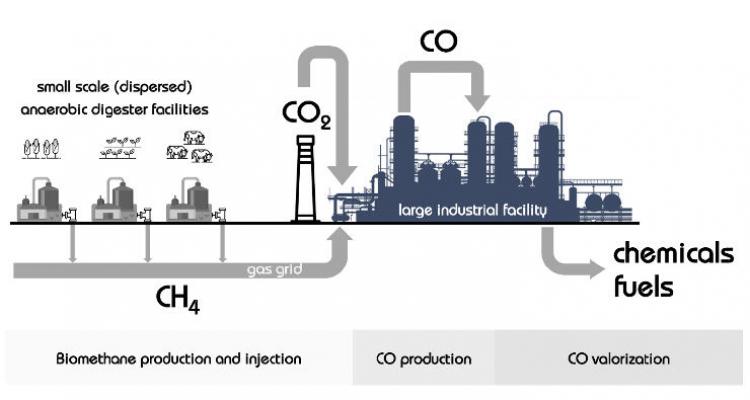
The biogas sector is rapidly growing and novel achievements lay the foundation for biogas plants as advanced bioenergy factories. Methane activation is one of the most challenging reactions to facilitate further conversion into higher molecules.
Chemical looping as a technology platform
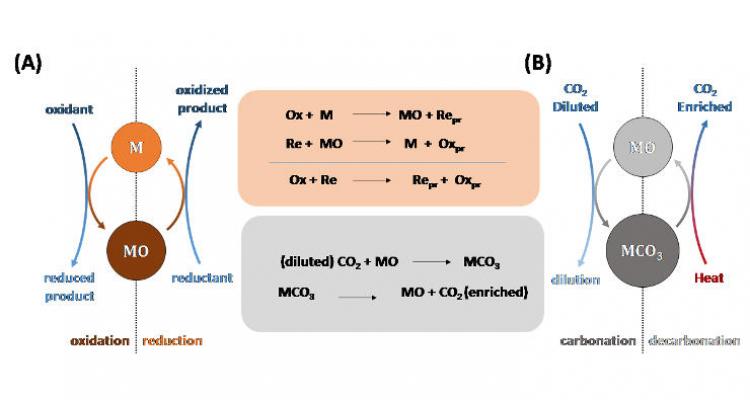
We aim to demonstrate that chemical looping has large potential, well beyond combustion, as a flexible technology platform for process intensification in fuel processing technology. The product separation that is inherent to this technology can have significant impact in the future on a broad range of chemical processes, with higher efficiency and ‘greener’ chemistry.
Multi-functional nanostructured materials
Next-generation material production aims at combining several functionalities to enable property flexibility. In combination with nanoscience unprecedented application potential can be achieved.
